Dear student,
The close packed structures have both octahedral and tetrahedral voids. In a ccp structure, there is 1 octahedral void in the centre of the body and 12 octahedral void on the edges. Each one of which is common to four other unit cells. Thus, in cubic close packed structure.
Octahedral voids in the centre of the cube =1
Effective number of octahedral voids located at the 12 edge of = 12 × 1/4 = 3
∴ Total number of octahedral voids = 4
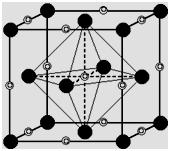
In ccp structure, there are 8 tetrahedral voids. In close packed structure, there are eight spheres in the corners of the unit cell and each sphere is in contact with three groups giving rise to eight tetrahedral voids
|

|
 |
|
Circles labelled T represent the centers of the tetrahedral interstices in the ccp arrangement of anions. The unit cell "owns" 8 tetrahedral sites.
Please feel free to ask your queries here. We are all IITians and here to help you in your IIT JEE preparation.
All the best.
Win exciting gifts by answering the questions on Discussion Forum. So help discuss any query on askiitians forum and become an Elite Expert League askiitian.
Now you score 5+15 POINTS by uploading your Pic and Downloading the Askiitians Toolbar respectively : Click here to download the toolbar..
Askiitians Expert
Sagar Singh
B.Tech, IIT Delhi
|
Circles labelled O represent centers of the octahedral interstices in the ccp arrangement of anions (fcc unit cell). The cell "owns" 4 octahedral sites.
|




