Dear jaisson
Nitrogen:
This molecule has ten electrons. The atomic orbitals combine to produce the following molecular orbital diagram

Here the 2pg orbital is occupied by two electrons to give a total bond order of three. This corresponds well with the Lewis structure (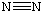 ), although the orbital approach tells us that there is one s and two p.
), although the orbital approach tells us that there is one s and two p.
Oxygen:
This molecule has twelve electrons, two more than nitrogen - and these extra two are placed in a pair of degenerate pg orbitals. The atomic orbitals combine to produce the following molecular orbital diagram:
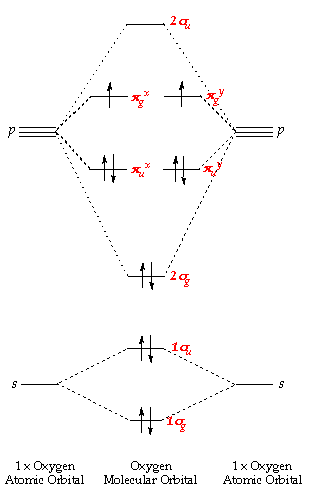
Comparison of the above energy level diagram wit hthat for nitrogen - you can see that the 2sg level lies lower than pu. Here, we are starting to fill the anti-bonding orbitals originating from the p orbital interactions and so the bond order decreases from three to two.
The lowest energy arrangment (Hund's rule) - has a single electron, each with parallel spins, in each of the pgx and pgy orbitals. This produces a paramagnetic molecule, with a double bond and has two unpaired electrons.
Please feel free to post as many doubts on our discussion forum as you can.
If you find any question Difficult to understand -
post it here and we will get you the answer and detailed solution very quickly.
We are all IITians and here to help you in your IIT JEE preparation.
All the best.
Regards,
Askiitians Experts
Badiuddin





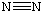 ), although the orbital approach tells us that there is one
), although the orbital approach tells us that there is one 





