According to the Van’t Hoff who study osmotic pressure of the dilute or ideal solutions,he suggested that a dilute or ideal solution behaves like an ideal gas and the different and the different gas laws are applicable to the dilute solutions as well.
For dilute solutions,Van’t Hoff observed,the osmotic pressure(
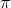
) is given as:--
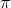
= cRT
where c = molar concentration of the solution ,
T = temperature and
R = gas constant
Thus for a solution,at a given temperature,both R and T are constant.
Tore,
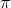
is directly proportional to c.Since osmotic pressure depends upon the molar concentration of the solution,therefore it is a colligative property.
As -- c = n/V
where n is the number of moles of solute dissolved in V litre of the solution,so
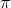
V = nRT
this is the equation called as Van’t Hoff equation for dilute solution.





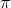 ) is given as:--
) is given as:--




