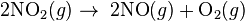Ask a Doubt
Get your questions answered by the expert for free
Enter text here...
Other Related Questions on physical chemistry

Which of the following aqueous solutions remains neutral afterelectrolysis
physical chemistry
0 Answer Available
Last Activity: 2 Years ago

In the reaction A + 2B ⇌ 2C, if 2 moles of A, 3 moles of B and 2 moles of C are placed in a 2 L flask andthe equilibrium concentration of C is 0.5 mol/L. The equilibrium constant (KC) for the reaction is.....and please tell how you know that the reaction is going forward .. imean how to calculate the reaction quotient ???
physical chemistry
1 Answer Available
Last Activity: 2 Years ago


18 19 gram mixture of na2co3 and nahco3 on heating gives 2.2 gram CO2 gas find the percent nahco3 (by mass) in mixture
physical chemistry
1 Answer Available
Last Activity: 2 Years ago







![\ -\frac{d[A]}{dt} = 2k[B]^2](http://upload.wikimedia.org/math/7/8/1/781cda9b25d2e92fb50c2f6a30188f41.png) or
or ![\ -\frac{d[A]}{dt} = k[A][B]](http://upload.wikimedia.org/math/c/f/d/cfd5938b3763344a202d6975a0d68ed9.png) or
or ![\ -\frac{d[A]}{dt} = 2k[A]^2](http://upload.wikimedia.org/math/8/7/3/873a3b7d216517de98cb8ee3a86ceb44.png)
![-\frac{d[A]}{dt} = k[A]^2](http://upload.wikimedia.org/math/5/8/c/58cd77b68b6d8db6df381fc176b25a1d.png) . Conflating the 2 inside the constant for the first, derivative, form will only make it required in the second, integrated form (presented below). The option of keeping the 2 out of the constant in the derivative form is considered more correct, as it is almost always used in peer-reviewed literature, tables of rate constants, and simulation software.
. Conflating the 2 inside the constant for the first, derivative, form will only make it required in the second, integrated form (presented below). The option of keeping the 2 out of the constant in the derivative form is considered more correct, as it is almost always used in peer-reviewed literature, tables of rate constants, and simulation software.![\frac{1}{[A]} = \frac{1}{[A]_0} + kt](http://upload.wikimedia.org/math/6/6/4/664842f28db997704b7ddbc61f6a7be8.png)
![\frac{[A]}{[B]} = \frac{[A]_0}{[B]_0} e^{([A]_0 - [B]_0)kt}](http://upload.wikimedia.org/math/0/2/6/0261d2c287f23e6971bba8e5cdf68b22.png)
![\ t_ \frac{1}{2} = \frac{1}{k[A]_0}](http://upload.wikimedia.org/math/9/c/e/9ce65475b614802ec855072f11df0d99.png) . For a second-order reaction half-lives progressively double.
. For a second-order reaction half-lives progressively double.![\ln{}r = \ln{}k + 2\ln\left[A\right]](http://upload.wikimedia.org/math/c/f/0/cf04309d2e7d76fc386ebfcf6877723f.png)