1. Exactly 0.200 mole electrons is passed through two electrolytic cells in series. One contains silver ion, and the other, zinc ion .assume that the only cathode reaction in each cell is the reduction of the ion to the metal. How many grams of each metal will be deposited? (at. Mass of zn = 65, ag = 108)
a. 108g ag and 65 g zn.
b. 108 ag and 32.5g zn.
c. 21.6 ag and 6.5 g zn.
d. 21.6 g ag and 13 g zn.
2. A steady current of 30.0A for 70.2 min corresponds to a passage of a. 1.2610^5 electrons.
b. 1.26×10^5 f.
c. 1.31 f.
d. 1.31 c.
3. Cu+ is not stable and undergoes disproportion. E for cu+ disproportionation.
(E° cu2+/cu =0.153V , E° cu+/cu=0.53V).
1. Exactly 0.200 mole electrons is passed through two electrolytic cells in series. One contains silver ion, and the other, zinc ion .assume that the only cathode reaction in each cell is the reduction of the ion to the metal. How many grams of each metal will be deposited? (at. Mass of zn = 65, ag = 108)
a. 108g ag and 65 g zn.
b. 108 ag and 32.5g zn.
c. 21.6 ag and 6.5 g zn.
d. 21.6 g ag and 13 g zn.
2. A steady current of 30.0A for 70.2 min corresponds to a passage of a. 1.2610^5 electrons.
b. 1.26×10^5 f.
c. 1.31 f.
d. 1.31 c.
3. Cu+ is not stable and undergoes disproportion. E for cu+ disproportionation.
(E° cu2+/cu =0.153V , E° cu+/cu=0.53V).





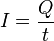
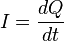 .
. 




