Dear nitin,
Lewis Structures for Covalent Compounds
- In a covalent compound, electrons are shared between atoms to form a covalent bond in order that each atom in the compound has a share in the number of electrons required to provide a stable, Noble Gas, electronic configuration.
- Electrons in the Lewis Structure (electron dot diagram) are paired to show the bonding pair of electrons.
- Often the shared pair of electrons forming the covalent bond is circled
- Sometimes the bond itself is shown (-), these structures can be referred to as valence structures.
Examples
hydrogen fluoride, HF
- Hydrogen atom has 1 valence electron

- Fluorine atom has 7 valence electrons
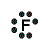
- Hydrogen will share its electron with fluorine to form a bonding pair of electrons (covalent bond) so that the hydrogen atom has a share in 2 valence electrons (electronic configuration of helium) and fluorine has a share in 8 valence electrons (electronic configuration of neon)
- Lewis Structure (electron dot diagram) for hydrogen fluoride
 OR
OR 
- Valence Structure for hydrogen fluoride

ammonia, NH3
- Nitrogen atom has 5 valence electrons

- Hydrogen atom has 1 valence electron

- Each of the 3 hydrogen atoms will share its electron with nitrogen to form a bonding pair of electrons (covalent bond) so that each hydrogen atom has a share in 2 valence electrons (electronic configuration of helium) and the nitrogen has a share in 8 valence electrons (electron configuration of neon)
- Lewis Structure (electron dot diagram) for ammonia
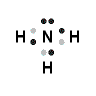 OR
OR 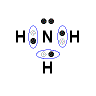
Please feel free to ask your queries here. We are all IITians and here to help you in your IIT JEE preparation.
All the best.
Win exciting gifts by answering the questions on Discussion Forum. So help discuss any query on askiitians forum and become an Elite Expert League askiitian.
Now you score 5+15 POINTS by uploading your Pic and Downloading the Askiitians Toolbar respectively : Click here to download the toolbar..
Askiitians Expert
Sagar Singh
B.Tech, IIT Delhi






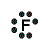
 OR
OR 


 OR
OR 





