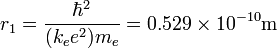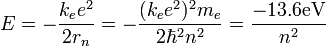assume that the potential energy of a hydrogen atom in its ground state is zero . then its energy in the first excited state will be
assume that the potential energy of a hydrogen atom in its ground state is zero . then its energy in the first excited state will be