 8 grade science> State BOYLE”S LAW....
8 grade science> State BOYLE”S LAW....

 14 Answers
14 Answersash jai
Raheema Javed
The absolute pressure exerted by a given mass of an ideal gas is inversely proportional to the volume it occupies if the temperature and amount of gas remain unchanged within a closed system.
Mathematically, Boyle's law can be stated as
P directly proportional to 1/V
or
PV = k
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as
P1 V1 = P2 V2.
The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases.
Mohammed Anas
Boyle's law (sometimes referred to as the Boyle–Mariotte law, or Mariotte's law is an experimental gas law which describes how the pressure of a gas tends to decrease as the volume of a gas increases. A modern statement of Boyle's law is:
The absolute pressure exerted by a given mass of an ideal gas is inversely proportional to the volume it occupies if the temperature and amount of gasremain unchanged within a closed system.
Mathematically, Boyle's law can be stated as
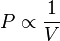
or
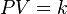
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Mohammed Anas
Boyle's law (sometimes referred to as the Boyle–Mariotte law, or Mariotte's law is an experimental gas law which describes how the pressure of a gas tends to decrease as the volume of a gas increases. A modern statement of Boyle's law is:
The absolute pressure exerted by a given mass of an ideal gas is inversely proportional to the volume it occupies if the temperature and amount of gasremain unchanged within a closed system.
Mathematically, Boyle's law can be stated as
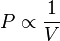
or
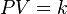
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Mohammed Anas
Boyle's law (sometimes referred to as the Boyle–Mariotte law, or Mariotte's law∂ is an experimental gas law which describes how the pressure of a gas tends to decrease as the volume of a gas increases. A modern statement of Boyle's law is:
The absolute pressure exerted by a given mass of an ideal gas is inversely proportional to the volume it occupies if the temperature and amount of gasremain unchanged within a closed system.
Mathematically, Boyle's law can be stated as
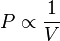
or
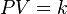
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Indranil Roy
Mathematically, Boyle's law can be stated as
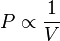
or
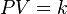
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Indranil Roy
Mathematically, Boyle's law can be stated as
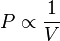
or
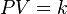
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Indranil Roy
Mathematically, Boyle's law can be stated as
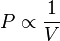
or
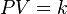
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Indranil Roy
Mathematically, Boyle's law can be stated as
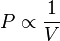
or
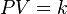
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Indranil Roy
Mathematically, Boyle's law can be stated as
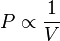
or
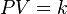
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Indranil Roy
Mathematically, Boyle's law can be stated as
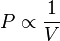
or
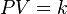
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Indranil Roy
Mathematically, Boyle's law can be stated as
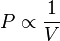
or
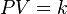
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Indranil Roy
Mathematically, Boyle's law can be stated as
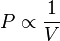
or
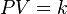
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.
Indranil Roy
Mathematically, Boyle's law can be stated as
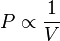
or
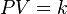
where P is the pressure of the gas, V is the volume of the gas, and k is a constant.
The equation states that product of pressure and volume is a constant for a given mass of confined gas as long as the temperature is constant. For comparing the same substance under two different sets of condition, the law can be usefully expressed as

The equation shows that, as volume increases, the pressure of the gas decreases in proportion. Similarly, as volume decreases, the pressure of the gas increases. The law was named after chemist and physicist Robert Boyle, who published the original law in 1662.

LIVE ONLINE CLASSES
Prepraring for the competition made easy just by live online class.

Full Live Access

Study Material

Live Doubts Solving

Daily Class Assignments
Other Related Questions on 8 grade science

I want complete notes on the chapter friction
In my book subtopics are:
Friction
Types of friction
Factors affecting friction
Methods of reducing friction
Methods of increasing friction
Advantages and disadvantages of friction
Some real life based situation where friction is applied.
I want complete notes on the chapter friction
In my book subtopics are:
Friction
Types of friction
Factors affecting friction
Methods of reducing friction
Methods of increasing friction
Advantages and disadvantages of friction
Some real life based situation where friction is applied.
8 grade science
Last Activity: 8 Months ago

How to make a monomer into polymer if it is an addition polymer example
How to make a monomer into polymer if it is an addition polymer example
8 grade science
Last Activity: 8 Months ago

This planet, _______ is yellowish in colour and has many rings and satellites.
A. Uranus
B. Jupiter
C. Neptune
D. Saturn
This planet, _______ is yellowish in colour and has many rings and satellites.
A. Uranus
B. Jupiter
C. Neptune
D. Saturn
8 grade science
Last Activity: 11 Months ago

Pasteurized milk does not need to be boiled. Why?
Pasteurized milk does not need to be boiled. Why?
8 grade science
Last Activity: 11 Months ago


