Ideal solutions :
An ideal solution one in which the molecules attract one another with equal force irrespective of their nature. Thus, a solution composed of two components A and B will be an ideal one if the forces between A and A, B and B should be the same. An ideal solution possesses the following characteristics:
(i) Volume change of mixing should be zero.
 ?Vmix = 0; Vsolvent + Vsolute = Vsolution
?Vmix = 0; Vsolvent + Vsolute = Vsolution
(ii)Heat change on mixing should be zero.
?Hmix = 0 (Heat is neither absorbed nor evolved).
(iii)There should be no chemical reaction between solvent and solute.
solvent + solute → solution
H2O + NH3 → NH4OH
H2O + CO2 → H2CO3 Non-ideal
H2O + CaO → Ca(OH)2
(iv)Solute molecules should be not dissociate in the ideal solution.
Aqueous medium
NaCl —————————→ Na+ + Cl−
Non-ideal
Aqueous medium
H2SO4 —————————→ 2H++ SO42–
(v)Solute molecules should not associate in ideal solution.
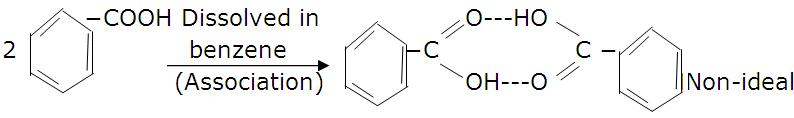
(vi)Ideal solutions must obey Raoult’s law at all concentrations.
The following are some of the binary mixtures which show the properties of ideal solutions.
(a) Benzene and toluene.
(b) Carbon tetrachloride, and silicon tetrachloride,
(c) N-Hexane and n-heptane,
Ethylene dibromide and tethylene dichloride.
To read more, Buy study materials of Solutions comprising study notes, revision notes, video lectures, previous year solved questions etc. Also browse for more study materials on Chemistry here.
View courses by askIITians


Design classes One-on-One in your own way with Top IITians/Medical Professionals
Click Here Know More

Complete Self Study Package designed by Industry Leading Experts
Click Here Know More

Live 1-1 coding classes to unleash the Creator in your Child
Click Here Know More
