Anomalous Properties of Lithium
Table of Content |
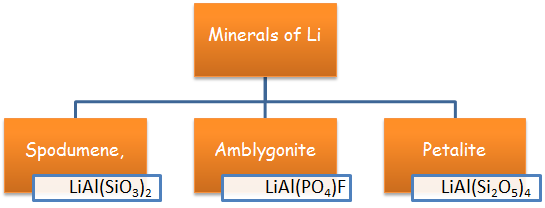
Introduction about Lithium
Group 1 elements of the periodic table are known as Alkali Metals. Lithium is the first element of the group 1. The small size of lithium allows it to behave differently from other alkali elements (Anomalous behavior).
Nature of Lithium
Lithium is highly electropositive in nature which allows it to form covalent bonds. The polarization behavior of lithium ion is comparable to that of magnesium ion. Thus, lithium shows a diagonal relationship with magnesium. There are various reasons for diagonal relationship of lithium with magnesium. These reasons are as follows:
-
As one moves from top to bottom in a group, electropositive nature increases. But from left to right in a period, electropositive nature decreases. Due to this, similar properties are observed between diagonal elements.
-
From top to bottom in a group, size increases; due to this polarizing power decreases but moving left to right in period polarizing power increases, thus diagonal elements show similar properties.
-
Lithium is a strong element similar to magnesium.
-
Lithium and magnesium ion show comparable boiling and melting points.
-
Due to small size, the lithium atom has high ionization energy. It reacts with water, liquid bromine and forms a highly stable hydride unlike other alkali metals. Magnesium exhibits all these properties.
Fig. 1. Reasons for Diagonal Relationship
-
Lithium and magnesium both forms monoxides.
2Mg + O2→ 2MgO
-
Lithium and magnesium both reacts with nitrogen to form lithium nitride and magnesium nitride.
(s) + N2(g) → 2Li3N(s)
-
Lithium and magnesium both reacts slowly with water and form oxides and hydroxides which decompose on heating.
Mg(s) + 2H2O(g) → Mg(OH)2(aq) + H2(g)
-
Oxides of both lithium and magnesium do not form superoxides.
-
The carbonates of lithium and magnesium decompose on heating to form the oxides and carbon-dioxide.
2 Mg(s) + CO2 2 MgO(s) + C(s)
-
Lithium chloride (LiCl) and magnesium chloride (MgCl2) both are soluble in ethanol.
-
Both LiCl and MgCl2 are deliquescent and form hydrates LiCl·2H2O and MgCl2·8H2O.
-
Both magnesium and lithium are less stable towards heat.
Uses of Lithium
Fig. 2. Uses of Lithium
Difference between Lithium and Other Alkali Metals
-
Lithium is harder than other alkali metals.
-
Melting and boiling point is higher than other alkali metals.
-
Lithium is least reactive out of all alkali metals.
-
It is a strong reducing agent compared to other alkali metal.
It is the only alkali metal that form the monoxide, Li2O.
4Li(s) + O2(g) → 2Li2O(s)
- Unlike other alkali metals, it forms Li3N.
6 Li + N2→ 2 Li3N
-
It does not form solid hydrogen carbonates compared to other alkali metals.
-
Lithium does not react with ethyne to form ethynide. But all other alkali metals form ethynide.
-
Lithium nitrate on heating form lithium monoxide as compared to other alkali metals which forms nitrites on decomposition.
4 LiNO3→ 2 Li2O + 4NO2 + O2
-
Lithium reacts slowly with bromine as compared to other alkali metals.
-
Lithium when heated in NH3 forms amide, Li2 NH while other alkali metals form amides, MNH2. M can be other alkali metals.
Watch this Video for more reference
More Readings
View courses by askIITians


Design classes One-on-One in your own way with Top IITians/Medical Professionals
Click Here Know More

Complete Self Study Package designed by Industry Leading Experts
Click Here Know More

Live 1-1 coding classes to unleash the Creator in your Child
Click Here Know More