X-Rays
Table of Content |
 X-rays were discovered accidentally by German scientist Rontgen in 1895. The first Nobel Prize was awarded to Rontgen in 1901. This highly penetrating electromagnetic radiation has proved to be a very powerful tool to study the crystal structure, in material research, in the radiography of metals and in medical sciences. Laue, Henry and Lawrence Bragg, Barkla and Siegbahn were some of the Nobel Laureates who have made contribution to this field. λ - ray spectroscopy and electron-spectroscopy were some of the spin-offs of these studies apart from the discovery of elements. X-ray is a electromagnetic spectrum.
X-rays were discovered accidentally by German scientist Rontgen in 1895. The first Nobel Prize was awarded to Rontgen in 1901. This highly penetrating electromagnetic radiation has proved to be a very powerful tool to study the crystal structure, in material research, in the radiography of metals and in medical sciences. Laue, Henry and Lawrence Bragg, Barkla and Siegbahn were some of the Nobel Laureates who have made contribution to this field. λ - ray spectroscopy and electron-spectroscopy were some of the spin-offs of these studies apart from the discovery of elements. X-ray is a electromagnetic spectrum.
Rontgen discovered that when the radiations called x-rays are passed through arms and hands or any other body part, they create detailed images of the inner bones. Whenever a person gets his x-ray done, an x-ray sensitive film is put on one side of the body and then the x-rays are shot through the person. While the skin is transparent, the bones are dense and absorb more x-rays. This is why the shadow of bones is left on the x-ray film while the skin does not appear in the x-ray. Below figure shows the wavelength of different electromagnetic spectrum.
X-rays are highly energetic radiations with very short wavelengths. Their wavelength is even shorter than the ultraviolet radiations and varies between 0.03 and 3 nanometers and some x-rays are as small as a single atom of many elements.
Working of X-Rays
X-rays are basically a type of radiations. They are also sources of energy like light but are slightly different in nature. Light has lower frequency than the x-rays and while it is absorbed by the skin, x-rays have higher frequency and pass through the human body. As the radiations of x-rays pass through the body, the energy particles called photons are absorbed at different rates. This whole pattern is depicted on x-ray images. The skin appears as transparent and so is not visible in the images. The dense parts like bones appear as white areas in the image whereas the softer parts like the heart and lungs are visible as darker areas.
How Safe are the X-Rays?
Being radiations, high levels of x-rays are obviously harmful. But the rays used for medical purposes are not harmful and are in fact very safe since the intensity and quantity of radiation is small. The intensity of radiation with regard to long term risk is measured in unit called millisieverts (mSv).
Experimental Production of X-Rays and the Bragg Spectrometer
Electrons from a heated element were accelerated by very high potential and made to impinge on the target (anode). The X-rays produced are collimated by parallel plates and are incident on a crystal (LiF, quartz, diamond, etc.) As the inter-atomic distance is of the same order as the wavelength of X-rays diffraction is produced and they are detected by counters or photographic plates.
Watch this Video for more reference
X-Ray Production
In an X-ray tube the electrons emitted from the cathode are accelerated towards the metal target anode by an accelerating voltage of typically 50 kV. The high energy electrons interact with the atoms in the metal target. Sometimes the electron comes very close to a nucleus in the target and is deviated by the electromagnetic interaction. In this process, which is called bremsstrahlung (braking radiation), the electron loses much energy and a photon (X-ray) is emitted. The energy of the emitted photon can take any value up to a maximum corresponding to the energy of the incident electron.
The high energy electron can also cause an electron close to the nucleus in a metal atom to be displaced. This vacancy is filled by an electron further out from the nucleus. The well defined difference in binding energy, characteristic of the material, is emitted as a monoenergetic photon. When detected this X-ray photon gives rise to a characteristic X-ray line in the energy spectrum
Properties
X-rays with short wavelengths which are strong, deeply penetrating and are highly destructive are called hard x-rays. The rays used for medicinal purposes are comparatively less penetrating and have longer wavelengths and are called soft x-rays. X-rays have a dual nature. We now discuss the various properties of these radiations:
-
They pass through materials more or less unchanged
-
They cannot be refracted
-
Electric and magnetic fields do not have any effect on these rays
-
These radiations ionize the surrounding air by discharging electrified bodies
-
They have short wavelength varying between 0.1 A° to 1 A°.
-
They are produced when a metal anode is bombarded by very high energy electrons.
-
They do not require any medium for propagation
-
X-rays cannot be focused on a single point
-
These radiations cannot be heard or smelt
-
They cause photoelectric emission.
-
Intensity of X – rays depends on number of electrons hitting the target.
-
Cut off wavelength or minimum wavelength, where v(in volts) is the p.d. applied to the tube λmin = 12400 / V A°.
- Continuous spectrum appears due to retardation of electrons.
Characteristic Equation
(a) Characteristics equation, eV = hfm
e = electron charge;
V = accelerating potential
fm = maximum frequency of X radiation
(b) Characteristic Spectrum due to transition of electron from higher to lower,
 ∝ (z-b)2 ,
∝ (z-b)2 ,

Which is the Moseley's Law.
Here,  is the wave number, b = 1 for Ka and b = 7.4 for La
is the wave number, b = 1 for Ka and b = 7.4 for La
Where b is Shielding factor (different for different series).
(c) Bragg's Law,

(θ = angle for max intensity)
(d) Binding energy = – [Total Mechanical Energy]
(e) Velocity of electron in nth orbit for hydrogen atom is c/137n, where c = speed of light.
(f) For x – rays,

(g) Series limit of series means minimum wave length of that series.
Problem (JEE Advanced)
A potential difference of 103 V is applied across an X-ray tube. The ratio of the de-Broglie wavelength of the incident electrons to the shortest wavelength of X-rays produced is
(a) 1/20
(b) 1/100
(c) 1
(d) 1/104
(e/m = 1.8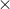 1014 C/kg for an electron)
1014 C/kg for an electron)
Solution:-
For incident electron
λ1 = h/p = h/√2Km = h/√ 2meV
and shortest wavelength of X-rays is λ2 = hc/Ve
Thus, λ1/λ2 = 1/c√(V/2) (e/m)
Substituting the values, we get, λ1/λ2 = 1
From the above observation we conclude that, option (c) is correct.


Question 1:-
In obtaining an X-ray photograph of our hand we use the principle of
(a) Photoelectric effect
(b) Ionization
(c) shadow photograph
(d) any of above
Question 2:-
X-rays are
(a) of unknown nature
(b) high energy electrons
(c) high energy photons
(d) radio isotopes
Question 3:-
Hydrogen atom does not emit X-rays because
(a) its energy levels are too close to each other
(b) its energy levels are too far apart
(c) it is too small in size
(d) it has a single electron
Question 4:-
In hydrogen spectrum which one of the following series lies in the ultraviolet region?
(a) Balmer series
(b) Pfund series
(c) Bracket series
(d) Lymann series
Question 5:-
Excited atoms return to their ground state in
(a) 10-10 s
(c) 10-6 s
(d) 10-9 s


| Q.1 | Q.2 | Q.3 | Q.4 | Q.5 |
|
c |
c |
d |
d |
b |
Related Resources
-
You might like to refer de Broglie Waves.
-
For getting an idea of the type of questions asked, refer the Previous Year Question Papers.
-
Click here to refer the most Useful Books of Physics.
- To get answer to any question related to X-rays click here.






