Atomic Structure Preparation Tips for IIT JEE
Table of the Content |
History of Atom
 It was the Greek philosopher Democritus (1460-370 BCE), who had a thought in his mind for first time ever in history that if we start breaking any matter in small particles continually, there will come a point from where it will not be possible to break it further into smaller particle i.e. matter cannot be infinitely divided. According to him, there must be an elementary particle which cannot be further divided and he called that elementary particle atom.
It was the Greek philosopher Democritus (1460-370 BCE), who had a thought in his mind for first time ever in history that if we start breaking any matter in small particles continually, there will come a point from where it will not be possible to break it further into smaller particle i.e. matter cannot be infinitely divided. According to him, there must be an elementary particle which cannot be further divided and he called that elementary particle atom.
But the theory of Democritus was rejected by two other well-known and more popular Greek Philosophers Aristotle and Plato. According to them matter was made up of four elements, air, water, fire and earth. As Aristotle and Plato were more popular than Democritus, their theory was well accepted by everyone.It was only after 2000 years that someone supported the theory of Democritus. Name of that person was John Dalton. He proposed a theory known as Dalton’s atomic theory.
The main postulates of Dalton’s Atomic Theory were
-
Matter is made up of small indivisible particles, called atoms.
-
Atoms can neither be created nor destroyed. This means that a chemical reaction is just a simple rearrangement of atoms and the same number of atoms must be present before and after the reaction.
-
Atom is the smallest particle of an element which takes part in a chemical reaction.
-
Atoms of the same element are identical in all respects especially, size, shape and mass.
-
Atoms of different elements have different mass, shape and size.
-
Atoms of different elements combine in a fixed ratio of small whole numbers to form compound atoms, called molecules.
The various topics that form a part of Atomic Structure include:
-
Sub-atomic Particles
-
Atomic models
-
Dual nature of Matter
-
Photoelectric Effect
-
Electromagnetic Radiations
-
Electronic Configuration
-
Discovery of Electron Proton and Neutron
You can get details explanation for these subtopics on the study material page of atomic structure.
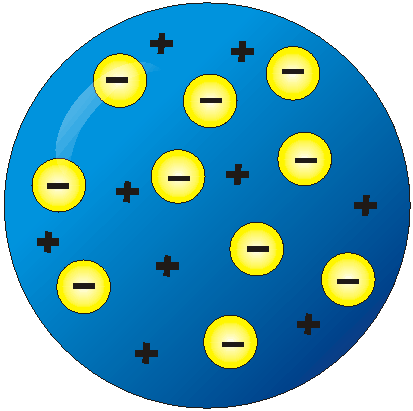
Atomic Models
Important Formulas
| Velocity of the electron |  |
| Radius of nth orbit |  |
| Wave length of EMR |  |
| Energy of photon |  |
| De Broglie equation |  |
| Threshold frequency |  |
How important is atomic structure for exams?
Atomic structure is a very important and comparatively easy to score topic. Weightage of this chapter in IIT JEE, NEET (AIPMT) and CBSE is about 2%, 3% and 8% of the total marks respectively.
What are the best books for the preparation of Atomic Structure?
Some of the books which are considered to be best for preparation of this section are:
NCERT text books are one of the best books for preparing any topic of chemistry for CBSE and NEET (AIPMT). For IIT JEE one should refer to other books like J.D Lee and O.P Tondon also.
Illustrations
|
Question 1: Energy of an electron is given by E = -2.178×10-18 J Z2/n2. Wavelength of light required to excite an electron in an hydrogen atom from level n =1 to n = 2 will be (h = 6.62 ×10−34Js and c = 3.0 ×108ms−1) A. 2.816 ×10−7 m B. 6.500 ×10−7m C. 8.500 ×10−7m D.1.214 ×10−7m Solution: |
How to Prepare Atomic Structure for IIT JEE?
It was mentioned earlier also, atomic structure is an easy and scoring chapter (specially for CBSE).Here are the following tips for preparing this chapter
-
First of all study this chapter from NCERT Text book. Read the chapter number of times and try to understand each and every
 concept clearly.
concept clearly. -
Take help of animation and videos which you will find easily on web, to understand the atomic models, photoelectric effect and other related topics. It is very important to have a clear picture of experimental setup and observations in your mind
-
Once you get all the concepts of chapter, move to the numeric problems.
-
Start with the solved examples given in the book and slowly move to the unsolved ones.
-
Once you solve all the questions given in NCERT Text book, you can move to the other books also and try to solve more problems given there.
-
After finishing the NCERT Text book, you can try J.D Lee and O.P Tondon also to gain in depth knowledge of the chapter.
-
Look into the previous year question papers for the questions related to the atomic structure and try to solve them all.
-
You will find that most of the numeric problems are from Behr’s atomic theory and hydrogen spectrum. These two topics are very important by exam. Try to solve as much questions as possible from these two topics.
To read more, Buy study materials of Structure of Atom comprising study notes, revision notes, video lectures, previous year solved questions etc. Also browse for more study materials on Chemistry here
View courses by askIITians


Design classes One-on-One in your own way with Top IITians/Medical Professionals
Click Here Know More

Complete Self Study Package designed by Industry Leading Experts
Click Here Know More

Live 1-1 coding classes to unleash the Creator in your Child
Click Here Know More