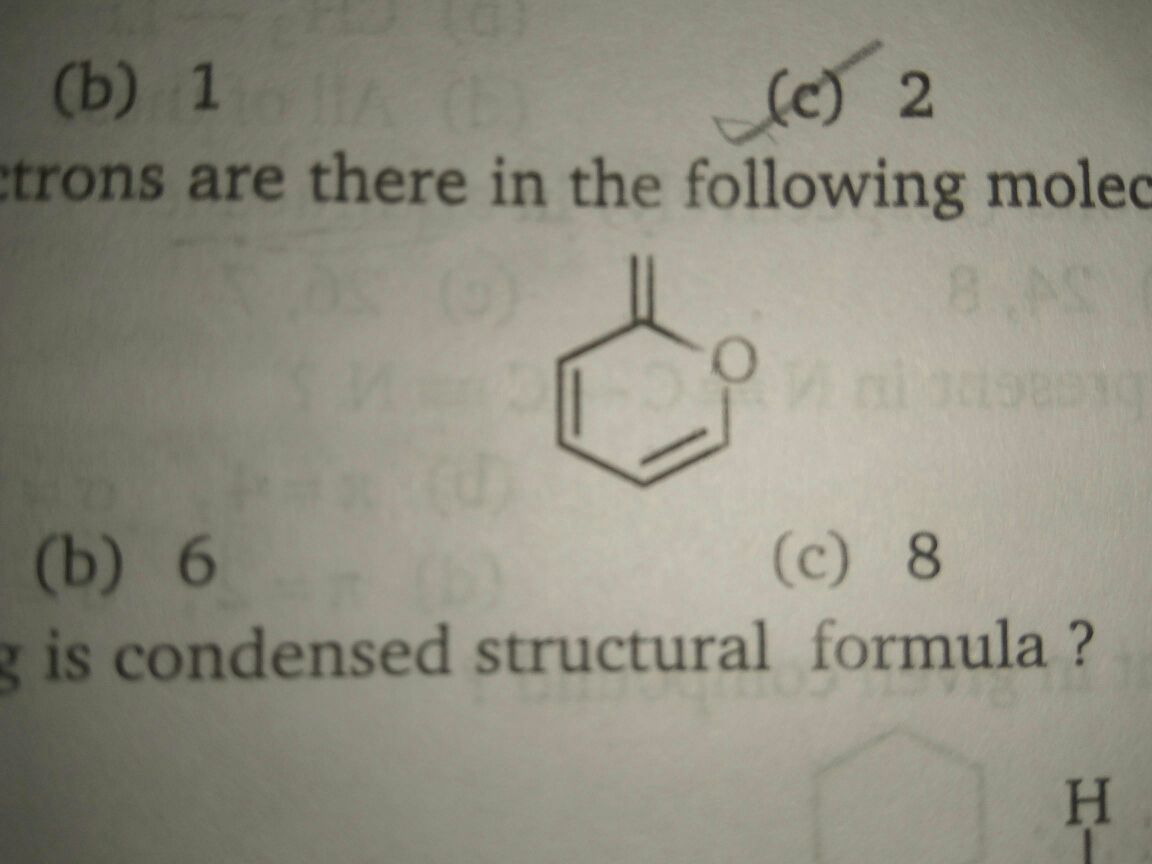
To address your question regarding pi electrons and the role of lone pairs on oxygen, let's break down the concepts step by step. Understanding the hybridization of oxygen and its electron configuration will clarify the relationship between lone pairs and pi electrons.
Understanding Hybridization and Electron Configuration
Oxygen, in its most common state, has an atomic number of 8, which means it has 8 electrons. The electronic configuration of oxygen is 1s² 2s² 2p⁴. In a typical sp² hybridization scenario, one of the 2s electrons is promoted to a 2p orbital, resulting in three hybrid orbitals (one s and two p orbitals) being formed. This configuration allows for the formation of sigma bonds and can also accommodate lone pairs.
Lone Pairs and Pi Electrons
In an sp² hybridized oxygen, one of the p orbitals remains unhybridized. This unhybridized p orbital is crucial because it can participate in forming pi bonds. However, it's essential to note that while we often refer to pi electrons in the context of double bonds, lone pairs can also participate in resonance structures, contributing to the overall electron delocalization within a molecule.
Analyzing the Compound
When looking at the compound in question, if oxygen is bonded in a way that allows it to contribute both its lone pairs to the overall electron cloud, these lone pairs can indeed be considered as part of the pi system, especially in conjugated systems or aromatic compounds. For example, in a compound like phenol, the lone pairs on the oxygen can delocalize and participate in resonance, effectively functioning as pi electrons.
- Example 1: In the case of phenol, the lone pair on the oxygen atom can overlap with the pi system of the aromatic ring, contributing to the total count of pi electrons.
- Example 2: In a conjugated system (like in certain carbonyl compounds), the lone pairs can also interact with adjacent pi bonds, enhancing the stability and reactivity of the molecule.
Calculating Pi Electrons
To find the total number of pi electrons in a compound, consider both the electrons in pi bonds and any electrons from lone pairs that can participate in resonance. If the compound has a structure that allows for delocalization, you might find that both lone pairs on the oxygen are counted as pi electrons, leading to the total of 10 pi electrons as noted in your question.
Summarizing the Key Points
In summary, although oxygen in an sp² hybridized state has a limited number of p orbitals, the role of lone pairs is essential in understanding pi electron contributions. They can participate in resonance and interact with pi systems, thus increasing the count of pi electrons in a compound. This concept is particularly relevant in larger molecules with delocalized electrons, where lone pairs contribute significantly to stability and reactivity.
So, when assessing pi electrons in a compound, remember to consider not just the double bonds but also how lone pairs can enhance the electron cloud through resonance and delocalization.