Other Related Questions on organic chemistry

'x' gm of a compound A3B2C5 contains 'y' gm of A atoms Using above informationMatch the following
'x' gm of a compound A3B2C5 contains 'y' gm of A atoms
Using above information
Match the following
organic chemistry
1 Answer Available
Last Activity: 3 Years ago

Dr. Ronald Lance is the greatest spell caster in the world also referred to as the most powerful saiki in the world by those who have used his powerful magic spells, weather you aiming at fixing your love, relationship or marriage, Ronald Lance is a real spells caster for your needs, bear in your mind that you are about to get in touch with our very experienced spells caster verified for casting authentic spells Call/Whats App+27634928462
Dr. Ronald Lance is the greatest spell caster in the world also referred to as the most powerful saiki in the world by those who have used his powerful magic spells, weather you aiming at fixing your love, relationship or marriage, Ronald Lance is a real spells caster for your needs, bear in your mind that you are about to get in touch with our very experienced spells caster verified for casting authentic spells Call/Whats App+27634928462
organic chemistry
3 Answers Available
Last Activity: 3 Years ago

Plz answer this query.....................................................................................................
Plz answer this query.....................................................................................................
organic chemistry
1 Answer Available
Last Activity: 3 Years ago

how to represent the possible fischer projections for 2-bromohexane
how to represent the possible fischer projections for 2-bromohexane
organic chemistry
1 Answer Available
Last Activity: 3 Years ago






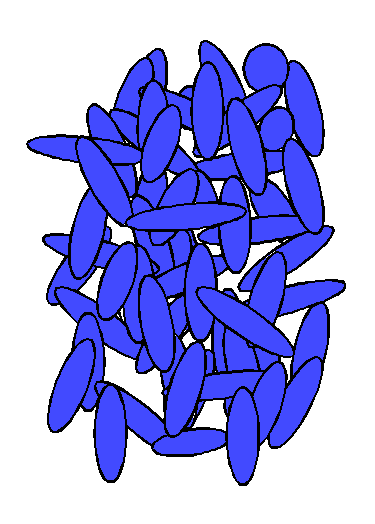
 To those who know that substances can exist in three states, solid, liquid, and gas, the term "liquid crystal" may be puzzling. How can a liquid be crystalline? However, "liquid crystal" is an accurate description of both the observed state transitions of many substances and the arrangement of molecules in some states of these substances.Many substances can exist in more than one state. For example, water can exist as a solid (ice), liquid, or gas (water vapor). The state of water depends on its temperature. Below 0°C, water is a solid. As the temperature rises above 0°C, ice melts to liquid water. When the temperature rises above 100°C, liquid water vaporizes completely. Some substances can exist in states other than solid, liquid, and vapor. For example, cholesterol myristate (a derivative of cholesterol) is a crystalline solid below 71°C. When the solid is warmed to 71°C, it turns into a cloudy liquid. When the cloudy liquid is heated to 86°C, it becomes a clear liquid. Cholesterol myristate changes from the solid state to an intermediate state (cloudy liquid) at 71°C, and from the intermediate state to the liquid state at 86°C. Because the intermediate state exits between the crystalline solid state and the liquid state, it has been called the liquid crystal state."Liquid crystal" also accurately describes the arrangement of molecules in this state. In the crystalline solid state, as represented in Figure 1, the arrangement of molecules is regular, with a regularly repeating pattern in all directions. (Molecules of substances with a liquid crystal state are generally oblong and rigid, that is, rod-shaped.) The molecules are held in fixed positions by intermolecular forces. As the temperature of a substance increases, its molecules vibrate more vigorously. Eventually, these vibrations overcome the forces that hold the molecules in place, and the molecules start to move. In the liquid state, this motion overcomes the intermolecular forces that maintain a crystalline state, and the molecules move into random positions, without pattern in location or orientation, as represented in Figure 2.In materials that form liquid crystals, the intermolecular forces in the crystalline solid are not the same in all directions; in some directions the forces are weaker than in other directions. As such a material is heated, the increased molecular motion overcomes the weaker forces first, but its molecules remain bound by the stronger forces. This produces a molecular arrangement that is random in some directions and regular in others. The arrangement of molecules in one type of liquid crystal is represented in Figure 3. The molecules are still in layers, but within each layer, they are arranged in random positions, although they remain more or less parallel to each other. Within layers, the molecules can slide around each other, and the layers can slide over one another. This molecular mobility produces the fluidity characteristic of a liquid.
To those who know that substances can exist in three states, solid, liquid, and gas, the term "liquid crystal" may be puzzling. How can a liquid be crystalline? However, "liquid crystal" is an accurate description of both the observed state transitions of many substances and the arrangement of molecules in some states of these substances.Many substances can exist in more than one state. For example, water can exist as a solid (ice), liquid, or gas (water vapor). The state of water depends on its temperature. Below 0°C, water is a solid. As the temperature rises above 0°C, ice melts to liquid water. When the temperature rises above 100°C, liquid water vaporizes completely. Some substances can exist in states other than solid, liquid, and vapor. For example, cholesterol myristate (a derivative of cholesterol) is a crystalline solid below 71°C. When the solid is warmed to 71°C, it turns into a cloudy liquid. When the cloudy liquid is heated to 86°C, it becomes a clear liquid. Cholesterol myristate changes from the solid state to an intermediate state (cloudy liquid) at 71°C, and from the intermediate state to the liquid state at 86°C. Because the intermediate state exits between the crystalline solid state and the liquid state, it has been called the liquid crystal state."Liquid crystal" also accurately describes the arrangement of molecules in this state. In the crystalline solid state, as represented in Figure 1, the arrangement of molecules is regular, with a regularly repeating pattern in all directions. (Molecules of substances with a liquid crystal state are generally oblong and rigid, that is, rod-shaped.) The molecules are held in fixed positions by intermolecular forces. As the temperature of a substance increases, its molecules vibrate more vigorously. Eventually, these vibrations overcome the forces that hold the molecules in place, and the molecules start to move. In the liquid state, this motion overcomes the intermolecular forces that maintain a crystalline state, and the molecules move into random positions, without pattern in location or orientation, as represented in Figure 2.In materials that form liquid crystals, the intermolecular forces in the crystalline solid are not the same in all directions; in some directions the forces are weaker than in other directions. As such a material is heated, the increased molecular motion overcomes the weaker forces first, but its molecules remain bound by the stronger forces. This produces a molecular arrangement that is random in some directions and regular in others. The arrangement of molecules in one type of liquid crystal is represented in Figure 3. The molecules are still in layers, but within each layer, they are arranged in random positions, although they remain more or less parallel to each other. Within layers, the molecules can slide around each other, and the layers can slide over one another. This molecular mobility produces the fluidity characteristic of a liquid.



