Hi
The electric field stores energy. The energy density of the electric field is given by

where
 is the permittivity of the medium in which the field exists
is the permittivity of the medium in which the field exists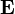 is the electric field vector.
is the electric field vector.
The total energy stored in the electric field in a given volume V is therefore

where
dV is the differential volume element
Energy associated with an electric field is 1/2ε0E2 . find the energy assosiated.
Then , apply the formula for photoelectric effect, i.e energy given = work fn + max K.E . Convert the respective units into ev.
Photoelectric effect
In effect quantitatively using Einstein's method, the following equivalent equations are used (valid for visible and ultraviolet radiation):
Energy of photon = Energy needed to remove an electron + Kinetic energy of the emitted electron
Algebraically:

where
- h is Planck's constant,
- f is the frequency of the incident photon,
- φ = hf0 is the work function (sometimes denoted W instead), the minimum energy required to remove a delocalised electron from the surface of any given metal,
 is the maximum kinetic energy of ejected electrons,
is the maximum kinetic energy of ejected electrons,- f0 is the threshold frequency for the photoelectric effect to occur,
- m is the rest mass of the ejected electron, and
- vm is the speed of the ejected electron.






 is the permittivity of the medium in which the field exists
is the permittivity of the medium in which the field exists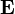 is the electric field vector.
is the electric field vector.

 is the maximum kinetic energy of ejected electrons,
is the maximum kinetic energy of ejected electrons,




