This is a question of chapter equilibrium. .. and i just want to know the formula used in it
This is a question of chapter equilibrium. .. and i just want to know the formula used in it
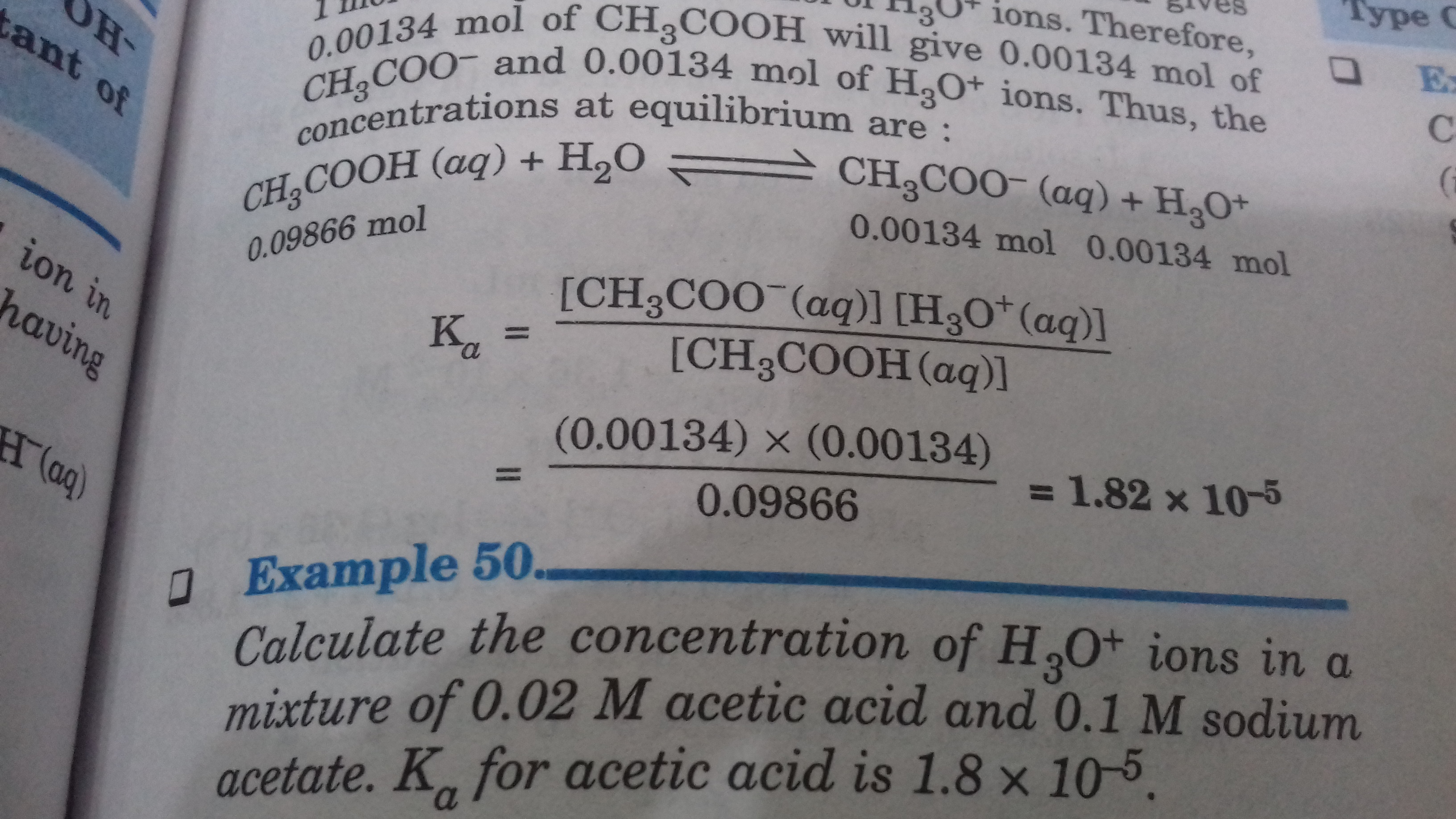

Last Activity: 3 Years ago

Last Activity: 3 Years ago

Last Activity: 3 Years ago


Last Activity: 3 Years ago