For reversible reactions, equilibrium contant K is related with temperature as
K = (A
f /A
r) e
–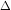 H / RT
H / RT , where A
f = Arrhenius constant of forward reaction, A
r = Arrhenius constant for reverse reaction.
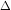
H = (E
a)
r – (E
a)
f = Difference of activation energies of reverse and forward reactions.
For endothermic reaction,
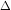
H > 0.
lnK = ln(A
f /A
r) + ln(e
–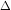 H / RT
H / RT) i.e. lnK = ln(A
f /A
r) –
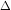
H / RT
lnK = (–
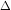
H / R)(1/T) + ln(A
f /A
r) ….(1). Compare (1) with y = mx + c
We infer from above equation that lnK vs 1/T graph is a straight line with negative slope (–
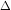
H / R).
Differentiate (1) wrt x.
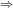
d(lnK)/dT = (–
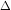
H / R)( –1/T
2) [

(–
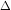
H / R) and ln(A
f /A
r) are constants.]
d(lnK)/dT =
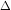
H / RT
2 > 0

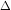
H, R, T
2 are positive.
If dT > 0, then d(lnK) >0 and if dT 0 i.e. if T is increased, K also increases.
This shows d(lnK) vs 1/T
2 graph will have positive slope [equation is d(lnK) = (
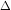
H / R)(dT) (1/T
2)].





