Ask a Doubt
Get your questions answered by the expert for free
Enter text here...
Other Related Questions on general physics

moment of inertia of a equilateral triangle about an axis passing through centre and perppendicular to the plane
general physics
1 Answer Available
Last Activity: 2 Years ago

. Because of the rotation of the earth, a plumb bob may not hang exactly along the direction ofthe earth’s gravitational pull (its weight) but deviates slightly from this direction. Calculate thedeviation (a) at 40 latitude, (b) at the poles, and (c) at the equator
general physics
1 Answer Available
Last Activity: 2 Years ago

The length of a massless rod is measured using vernier callipers having 10 vernier scale divisions and least count of 0.1 mm. Two masses 1.00 ± 0.01 kg and 4.00 ± 0.02 kg are attached to its ends. If reading of vernier scale is 5 divisions coinciding with 1.3 cm, what is the % error in locating centre of mass from lighter mass
general physics
2 Answers Available
Last Activity: 2 Years ago

A block is kept on x-y plane at a point (2m,3m). It is pushed with a force of magnitude 10 Ntowards point (5m,7m). The force can be represented as?
general physics
0 Answer Available
Last Activity: 2 Years ago

The phase of particle at t is 70°and another particle is at t is 790° what is wavelength if the distance between two particles is1 m
general physics
0 Answer Available
Last Activity: 2 Years ago





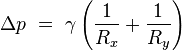
 is surface tension.
is surface tension.



