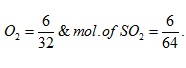1) Two containers P and Q of equal volume (1 litre each) contain 6 g of O2 and SO2 respectively at 300 K and 1 atmosphere, then
a) Number of molecules in P is less than that in Q
b) Number of molecules in P and Q is same
c) Number of molecules in Q is less than that in P
d) Either (a) or (b)
1) Two containers P and Q of equal volume (1 litre each) contain 6 g of O2 and SO2 respectively at 300 K and 1 atmosphere, then
a) Number of molecules in P is less than that in Q
b) Number of molecules in P and Q is same
c) Number of molecules in Q is less than that in P
d) Either (a) or (b)